RELATED PRODUCTS NOT AVAILABLE
Canada (FR)
true

- Overview
- Products & Accessories
- EIFU & Resources
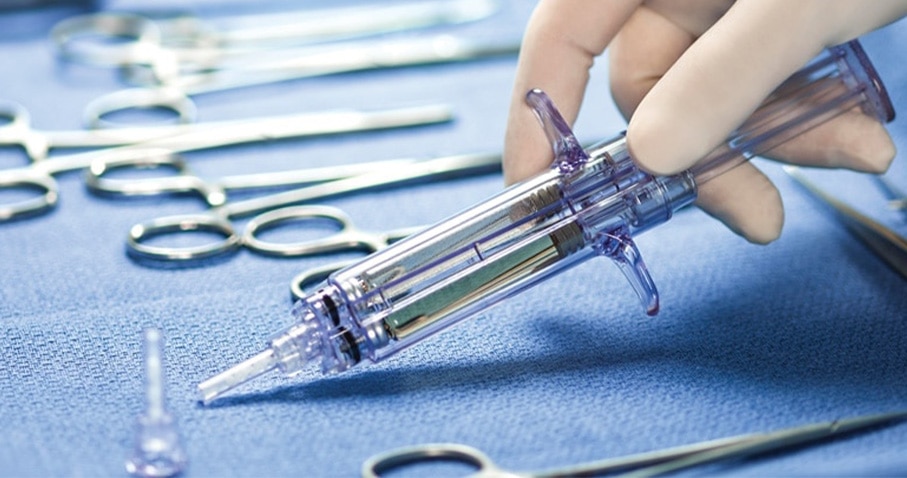
A strong elastic seal where it matters most
Tridyne™ Vascular Sealant is the latest addition to the BD family of hemostasis solutions, offering cardiovascular, cardiothoracic, and vascular surgeons a unique, easy-to-use solution to reinforce aortic anastomoses and control bleeding when adjunctive measures are required. By combining a proprietary formulation of polyethelyne glycol and human serum albumin, it forms a strong, flexible seal, even in anticoagulated patients.1,2 The Tridyne™ hydrogel is designed to adhere where it should – on both tissue and grafts.*
Preclinical data demonstrates that Tridyne™ Vascular Sealant resorbs naturally to maintain its seal strength through the critical postoperative period without compromising tissue healing.2
true
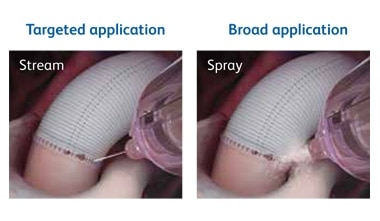
Precise, controlled application—made easy
Tridyne™ Vascular Sealant provides application control without the need for pressurized gas.
The Tridyne™ applicator is designed to:
- Provide uniform, continuous coverage without clumping
- Control sealant application by varying hand pressure
- Easily transition from targeted application over the anastomosis to broader coverage to address more diffuse bleeding
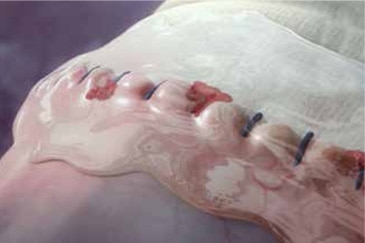
Clarity and coverage
The specialized Tridyne™ formulation is transparent and forms a completely clear hydrogel at the application site. With its unique combination of clarity and coverage, Tridyne™ Vascular Sealant allows for visual confirmation of hemostasis at the anastomotic suture line—making it an ideal choice for aortic surgery.

Performance under pressure
In a prospective, randomized, controlled multicenter study, Tridyne™ Vascular Sealant outperformed Gelfoam® Plus Hemostasis Kit in several critical measures. The study included 156 patients from 18 different centers who underwent thoracic aortic surgery, with hemostasis evaluated at the aortic anastomotic suture line.1
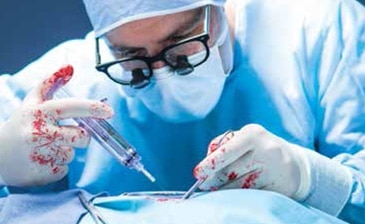
The proven solution during aortic repair1
Tridyne™ Vascular Sealant offers surgeons a specialized, easy-to-use solution during aortic repair, with unique application control and clinically proven results.1 With its demonstrated ability to provide fast, effective control of bleeding, it’s clearly the sealant choice for aortic surgery.
* The use of Tridyne™ Vascular Sealant with ePTFE grafts or vascular patches has not been studied clinically. Preclinical data on file. Preclinical test results may not correlate to clinical performance.
1. Khoynezhad A, DelaRosa J, Moon M, et al. Facilitating Hemostasis After Proximal Aortic Surgery: Results of The PROTECT Trial. Ann Thorac Surg. 2018;105(5):1357-1364.
2. BD Inc. In vitro testing. Data on file. Preclinical test results may not correlate to clinical performance.
3. Tridyne™ Vascular Sealant is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
4. One subject from the Gelfoam® Plus treatment group required a reoperation for aortic bleeding complications following completed surgery through 30 days.
5. Mean time to hemostasis was 2.07 minutes for the Tridyne™ Vascular Sealant treatment group and 6.3 minutes for the Gelfoam® Plus control treatment group (P < 0.0001).
Indications
Tridyne™ Vascular Sealant (Tridyne™ VS) is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
Contraindications
Tridyne™ VS is not for intravascular use. Do not use Tridyne™ VS in patients who have a history of allergic reaction to Human Serum Albumin, Polyethylene Glycol, or other device components. Do not apply Tridyne™ VS on oxidized regenerated cellulose, absorbable gelatin sponges, or any other surface or material other than the target tissue or graft as adherence and intended outcome may be compromised. Do not use Tridyne™ VS in patients who have insufficient renal capacity for clearance of the Polyethylene Glycol load.
Warnings
Do not use Tridyne™ VS as a substitute for standard closure techniques. Excessive pressure against the vessel/graft edges or surrounding tissue during application may result in separation of the vessel/graft edges, allowing sealant into the vessel.
Do not apply Tridyne™ VS when the vessel lumen is under negative pressure to avoid product from being drawn into the vessel (e.g., avoid applying in vessels that are not pressurized).
Do not use more than 30 mL of Tridyne™ VS per patient. Do not allow non-polymerized Tridyne™ VS to contact circulating blood.
Adverse Events
In a pivotal clinical trial, approximately half of the subjects in each treatment group experienced a Serious Adverse Event while in the study (51 subjects [48.1%] treated with Tridyne™ VS, 29 subjects [58.0%] treated with Control). Three Serious Adverse Events in the treatment group were considered by the Clinical Events Committee to be possibly device-related. These Serious Adverse Events included: cerebrovascular accident and serosanguineous pericardial effusion. None of the Serious Adverse Events were unexpected given the procedures performed. The details of these clinical trial adverse events can be reviewed in the Instructions for Use supplied with the product and are also available at www.bd.com. Caution: Federal (USA) law restricts this device to sale by or on order of a licensed physician or properly licensed practitioner.
true
BD-14650
* The use of Tridyne™ Vascular Sealant with ePTFE grafts or vascular patches has not been studied clinically. Preclinical data on file. Preclinical test results may not correlate to clinical performance.
1. Khoynezhad A, DelaRosa J, Moon M, et al. Facilitating Hemostasis After Proximal Aortic Surgery: Results of The PROTECT Trial. Ann Thorac Surg. 2018;105(5):1357-1364.
2. BD Inc. In vitro testing. Data on file. Preclinical test results may not correlate to clinical performance.
3. Tridyne™ Vascular Sealant is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
4. One subject from the Gelfoam® Plus treatment group required a reoperation for aortic bleeding complications following completed surgery through 30 days.
5. Mean time to hemostasis was 2.07 minutes for the Tridyne™ Vascular Sealant treatment group and 6.3 minutes for the Gelfoam® Plus control treatment group (P < 0.0001).
Indications
Tridyne™ Vascular Sealant (Tridyne™ VS) is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
Contraindications
Tridyne™ VS is not for intravascular use. Do not use Tridyne™ VS in patients who have a history of allergic reaction to Human Serum Albumin, Polyethylene Glycol, or other device components. Do not apply Tridyne™ VS on oxidized regenerated cellulose, absorbable gelatin sponges, or any other surface or material other than the target tissue or graft as adherence and intended outcome may be compromised. Do not use Tridyne™ VS in patients who have insufficient renal capacity for clearance of the Polyethylene Glycol load.
Warnings
Do not use Tridyne™ VS as a substitute for standard closure techniques. Excessive pressure against the vessel/graft edges or surrounding tissue during application may result in separation of the vessel/graft edges, allowing sealant into the vessel.
Do not apply Tridyne™ VS when the vessel lumen is under negative pressure to avoid product from being drawn into the vessel (e.g., avoid applying in vessels that are not pressurized).
Do not use more than 30 mL of Tridyne™ VS per patient. Do not allow non-polymerized Tridyne™ VS to contact circulating blood.
Adverse Events
In a pivotal clinical trial, approximately half of the subjects in each treatment group experienced a Serious Adverse Event while in the study (51 subjects [48.1%] treated with Tridyne™ VS, 29 subjects [58.0%] treated with Control). Three Serious Adverse Events in the treatment group were considered by the Clinical Events Committee to be possibly device-related. These Serious Adverse Events included: cerebrovascular accident and serosanguineous pericardial effusion. None of the Serious Adverse Events were unexpected given the procedures performed. The details of these clinical trial adverse events can be reviewed in the Instructions for Use supplied with the product and are also available at www.bd.com. Caution: Federal (USA) law restricts this device to sale by or on order of a licensed physician or properly licensed practitioner.
true
BD-14650
Literature
BD's collection of literature on industry and on our offerings gives you information you can use to continue striving for excellence.
Learn more
Training
BD offers training resources to help improve your clinical practices as part of our goal of advancing the world of health.
Learn more
Events
BD supports the healthcare industry with market-leading products and services that aim to improve care while lowering costs. We host and take part in events that excel in advancing the world of health™.
Learn more
Case Studies
BD promotes clinical excellence by providing various resources on best practices, clinical innovations and industry trends in healthcare.
Learn more
* The use of Tridyne™ Vascular Sealant with ePTFE grafts or vascular patches has not been studied clinically. Preclinical data on file. Preclinical test results may not correlate to clinical performance.
1. Khoynezhad A, DelaRosa J, Moon M, et al. Facilitating Hemostasis After Proximal Aortic Surgery: Results of The PROTECT Trial. Ann Thorac Surg. 2018;105(5):1357-1364.
2. BD Inc. In vitro testing. Data on file. Preclinical test results may not correlate to clinical performance.
3. Tridyne™ Vascular Sealant is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
4. One subject from the Gelfoam® Plus treatment group required a reoperation for aortic bleeding complications following completed surgery through 30 days.
5. Mean time to hemostasis was 2.07 minutes for the Tridyne™ Vascular Sealant treatment group and 6.3 minutes for the Gelfoam® Plus control treatment group (P < 0.0001).
Indications
Tridyne™ Vascular Sealant (Tridyne™ VS) is indicated for use in aortic surgery when adjunctive measures to achieve hemostasis are required by mechanically sealing areas of leakage.
Contraindications
Tridyne™ VS is not for intravascular use. Do not use Tridyne™ VS in patients who have a history of allergic reaction to Human Serum Albumin, Polyethylene Glycol, or other device components. Do not apply Tridyne™ VS on oxidized regenerated cellulose, absorbable gelatin sponges, or any other surface or material other than the target tissue or graft as adherence and intended outcome may be compromised. Do not use Tridyne™ VS in patients who have insufficient renal capacity for clearance of the Polyethylene Glycol load.
Warnings
Do not use Tridyne™ VS as a substitute for standard closure techniques. Excessive pressure against the vessel/graft edges or surrounding tissue during application may result in separation of the vessel/graft edges, allowing sealant into the vessel.
Do not apply Tridyne™ VS when the vessel lumen is under negative pressure to avoid product from being drawn into the vessel (e.g., avoid applying in vessels that are not pressurized).
Do not use more than 30 mL of Tridyne™ VS per patient. Do not allow non-polymerized Tridyne™ VS to contact circulating blood.
Adverse Events
In a pivotal clinical trial, approximately half of the subjects in each treatment group experienced a Serious Adverse Event while in the study (51 subjects [48.1%] treated with Tridyne™ VS, 29 subjects [58.0%] treated with Control). Three Serious Adverse Events in the treatment group were considered by the Clinical Events Committee to be possibly device-related. These Serious Adverse Events included: cerebrovascular accident and serosanguineous pericardial effusion. None of the Serious Adverse Events were unexpected given the procedures performed. The details of these clinical trial adverse events can be reviewed in the Instructions for Use supplied with the product and are also available at www.bd.com. Caution: Federal (USA) law restricts this device to sale by or on order of a licensed physician or properly licensed practitioner.
true
BD-14650
true